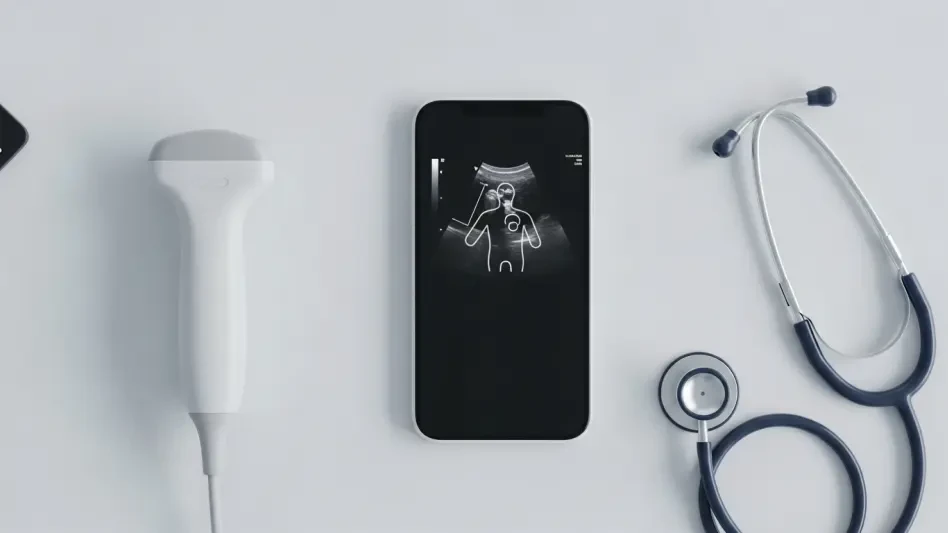
The long-standing challenge of medical imaging has never been the portability of the hardware, but rather the immense cognitive load required to interpret the resulting “snowstorm” of grayscale pixels. While high-end hospital carts have dominated the clinical landscape for decades, a new generation of pocket-sized devices is finally bridging the gap between raw data collection and actionable diagnosis. By embedding sophisticated artificial intelligence directly into the handheld workflow, these tools are transforming the stethoscope of the future into a visual powerhouse that does not require a decade of radiological training to operate effectively.
Evolution of Point-of-Care Ultrasound and AI Integration
The shift toward point-of-care ultrasound (POCUS) has been driven by the need for immediate bedside answers, yet traditional handheld units often left general practitioners struggling with image orientation. Early iterations of portable probes prioritized miniaturization and battery life, frequently at the expense of image clarity. This created a paradoxical situation where a physician could afford the device but lacked the specialized “ultrasound eye” to make it useful in a high-stakes clinical environment.
Modern integration of machine learning has fundamentally altered this trajectory by moving beyond simple image capture. Contemporary systems now serve as a digital co-pilot, contextualizing the live feed against vast libraries of anatomical data. This evolution is not merely about making the hardware smaller; it is about decentralizing expertise. By shifting the “intelligence” from the human brain to the software layer, the technology is moving ultrasound from the specialized radiology suite directly into the primary care exam room.
Key Technical Components of AI-Assisted Imaging
Real-Time Anatomical Recognition and T-Mode Visualization
One of the most disruptive technical leaps in this sector is the implementation of synchronized visualization modes, such as the T-Mode interface. This system functions by utilizing a split-screen display where one side shows the raw, unadulterated ultrasound signal while the other provides a color-coded, labeled digital twin. This dual-layer approach allows the user to see through the “noise” of biological tissue, using AI to highlight specific structures like the carotid artery or the meniscus in real time.
The performance of this system relies on low-latency processing that keeps the digital overlay perfectly mapped to the physical probe movements. Unlike static reference guides, this dynamic labeling adjusts as the physician tilts or slides the transducer. This technical capability is unique because it provides immediate feedback on whether the user has captured the correct plane, effectively reducing the “search time” that typically plagues novice sonographers.
Intelligent Interpretation Layers for Novice Users
Beyond mere recognition, these devices incorporate interpretation layers that act as a translation service for clinical findings. For instance, when scanning a heart, the software does not just identify the left ventricle; it can automatically calculate ejection fractions or detect wall motion abnormalities. These layers utilize neural networks trained on millions of confirmed clinical cases, providing a level of consistency that even experienced human operators can sometimes struggle to maintain during a busy shift.
This implementation is unique compared to traditional “auto-gain” features because it offers qualitative guidance rather than just quantitative adjustments. Instead of simply making the picture brighter, the AI suggests where to move the probe to optimize the view. This interactive loop transforms the device from a passive camera into an active instructor, which is essential for maintaining diagnostic standards in decentralized medical settings.
Current Advancements in Diagnostic Support Systems
The current landscape is shifting toward specialized diagnostic modules that target high-frequency clinical complaints. Recent updates have introduced dedicated modes for cardiac and orthopedic assessments, specifically designed to tackle the “gray areas” of general medicine. These advancements allow for automated measurement of joint gaps in the knee or the detection of fluid around the lungs, tasks that previously required a formal referral to a specialist imaging center.
Moreover, the integration of cloud-based collaboration allows these handheld units to transmit AI-flagged clips to remote specialists for instant second opinions. This hybrid model of AI-assisted local scanning and remote human verification represents a significant trend in tele-health. It ensures that the speed of a handheld device is matched by the accuracy of a specialized review, creating a more robust safety net for both the provider and the patient.
Clinical Applications in Primary and Specialized Care
In the realm of primary care, the ability to perform a rapid cardiac screen during a routine physical can lead to the early detection of heart failure. Patients presenting with vague symptoms like fatigue or mild shortness of breath often wait weeks for an echocardiogram. With AI-guided tools, a family physician can now visualize heart valves and chamber sizes within minutes, potentially initiating life-saving treatments or specialized referrals much earlier in the disease progression.
In specialized fields like sports medicine or physical therapy, these devices are used to visualize musculoskeletal injuries in real-time. By providing “textbook-style” replicas of the knee or shoulder anatomy, the technology allows therapists to show patients the exact site of a tear or inflammation. This visual confirmation not only improves diagnostic accuracy but also increases patient compliance, as the individual can see the internal rationale for their prescribed rehabilitation program.
Implementation Barriers and Technical Limitations
Despite these leaps, several hurdles remain, particularly regarding the “black box” nature of AI algorithms. Regulatory bodies and skeptical clinicians often question the reliability of automated interpretations, fearing that a false negative from the AI could lead to missed diagnoses. There is also the challenge of ergonomic fatigue; while the probes are small, maintaining the precise angle required for a high-quality scan still demands a level of manual dexterity that software cannot entirely replace.
Technical limitations also exist in terms of processing power and heat dissipation. Running complex neural networks on a battery-powered device causes significant thermal load, which can lead to shortened scan times or frame-rate drops. Furthermore, while the AI is excellent at recognizing standard anatomy, it may struggle with patients who have atypical physical structures or significant scarring, highlighting the ongoing need for human oversight and continuous software refinement.
The Future of Democratized Medical Imaging
The trajectory of this technology points toward a future where medical imaging is as ubiquitous as digital thermometry. As processing chips become more efficient and AI models more refined, the cost of these devices will likely continue to drop, making them accessible to rural clinics and under-resourced regions globally. This democratization will shift the focus of healthcare from reactive treatment to proactive monitoring, as routine screenings become a standard part of every patient encounter.
Looking ahead, we can expect the integration of predictive analytics into these handheld units. Instead of just identifying a current condition, the AI might analyze tissue texture or blood flow patterns to predict future risks, such as the likelihood of a stroke or the progression of chronic kidney disease. This shift from diagnostic tool to prognostic engine will redefine the role of the general practitioner, making them the primary gatekeepers of advanced physiological data.
Conclusion and Final Assessment
The review of AI-guided handheld ultrasound revealed a technology that successfully addressed the historical barriers to point-of-care imaging. By prioritizing the interpretation layer through T-Mode visualization and automated diagnostic support, these devices moved beyond the limitations of traditional hardware. The clinical evidence showed that primary care providers could now perform complex scans with a level of confidence previously reserved for specialists. While technical hurdles regarding processing heat and algorithmic transparency were noted, the overall impact on diagnostic speed and patient outcomes appeared overwhelmingly positive. Stakeholders should now focus on establishing standardized training protocols and integrating these AI insights directly into electronic health records to maximize the longitudinal value of every scan.